Abstract
Introduction: Peripheral blood samples of healthy individuals may harbour detectable mutations in genes recurrently mutated in myeloid malignancies, a situation referred to as clonal hematopoiesis (CH). Risk factors for CH include increasing age as well as previous exposure to cytotoxic therapy. CH has been associated with an increased risk of overall mortality, including in the setting of autologous stem cell transplantation (ASCT) for non-Hodgkin lymphoma (Gibson et al, JCO, 2017). The excess mortality is largely driven by cardio-vascular disease, but may also be additionally attributable to an increased risk of myeloid malignancies that arise through the selection of CH subclones. Herein, we aimed to investigate the prognostic implications of CH after ASCT in an independent and diversified, large cohort of lymphoma patients using ultra-deep, highly sensitive error-correction sequencing.
Methods: DNA was obtained from 420 residual apheresis products obtained from patients who had undergone autologous stem cell transplantation for lymphoma at the Princess Margaret Cancer Center between 2002 and 2018. Target DNA sequences corresponding to regions recurrently mutated in myeloid neoplasms (affecting n = 36 genes) were captured using single molecule molecular inversion probes (smMIPs) that incorporate molecular tagging. Single nucleotide variants and short insertions and deletions were identified using SmMIP-tools (Medeiros et al, bioRxiv, 2021), which implements a series of steps including probabilistic modeling of allele-specific error rates and generation of consensus sequences to suppress next-generation sequencing-associated errors. Given the high sensitivity and precision of our method, we did not prespecify a variant allele fraction cut-off.
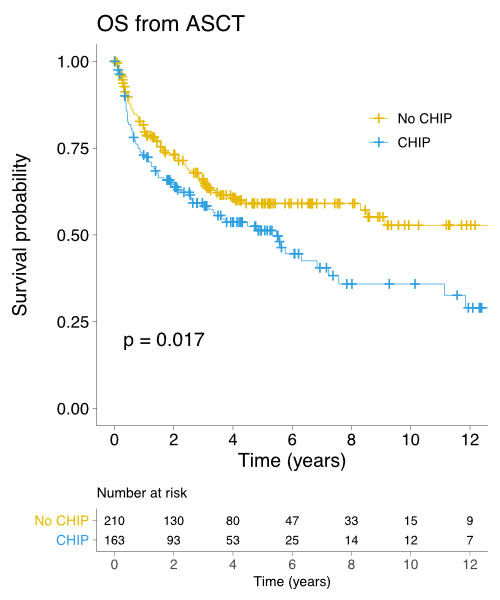
Results: All patients had relapsed/refractory lymphoma, except for 98 (23.3%) mantle cell lymphoma patients and one patient with extranodal NK/T-cell lymphoma where ASCT was part of frontline management. The most common conditioning regimens were high-dose melphalan and etoposide (77.5%) and high-dose melphalan and Ara-C (16.4%). We identified 275 high-confidence mutations in 181 out of 420 patients (43.1%), with 64 of these 181 patient samples (35.4%) having more than one mutation. The median age was higher in patients with CH than in patients without (55 years vs. 51, P = 0.002). The most frequently mutated gene were PPM1D (11.9%), followed by TET2 (11.4%), DNMT3A (8.8%), ASXL1 (5.2%) and TP53 (4.5%). The lymphoma subtype with the highest prevalence of CH was T-cell lymphoma (CH found in 72.2% of cases), followed by transformed indolent lymphoma (51.4%), mantle cell lymphoma (47.5%), diffuse large B-cell lymphoma (40.4%) and Hodgkin lymphoma (33.3%). While there was no difference in the number of CD34+ cells infused for patients with and without CH, the median time to neutrophil engraftment and the median time to platelet engraftment were significantly longer in patients with CH (11 days vs. 10 days, P = 0.025; and 14 days vs. 13 days, P < 0.001, respectively). The median follow-up of living patients was 4.2 years. Patients with CH had inferior 5-year OS from the time of first relapse (38.9% vs. 45.5%, P = 0.037) and from the time of ASCT (51.2% vs. 59.1%, P = 0.017, see figure). Five-year OS from ASCT was 47.5% vs. 53.7% in patients with 1 mutation and > 1 mutation, respectively, compared to 59.1% in patients without CH (P = 0.005). The presence of CH did not have an impact on the risk of post-ASCT relapse. In multivariate Cox regression analysis in which CH and age (as a continuous variable) were included, CH remained significantly associated with adverse OS post-ASCT (HR 1.39, 95% 1.02-1.91, P = 0.038). Only seven patients out of 420 (1.7%) developed a therapy-related myeloid neoplasm (TMN). The cumulative incidence of TMN was not significantly increased in patients with CH (10-year cumulative incidence 3.3% vs. 3.0% in those without CH, P = 0.433).
Conclusions: Our results show that CH was associated with delayed neutrophil and platelet engraftment. Moreover, CH conferred an increased risk of death after ASCT that was not explained by lymphoma relapse. The risk of TMN was low in our cohort and CH was not a risk factor for TMN, an observation that is distinct from prior observations (e.g. Gibson et al, JCO, 2017 and Husby et al, Leukemia, 2020). Our results raise the possibility that the risk of TMN may be modulated by factors other than CH.
Minden: Astellas: Consultancy. Kuruvilla: Janssen: Honoraria, Research Funding; Antengene: Honoraria; AstraZeneca: Honoraria, Research Funding; Amgen: Honoraria; Incyte: Honoraria; Novartis: Honoraria; Karyopharm: Honoraria, Other: Data and Safety Monitoring Board; Pfizer: Honoraria; AbbVie: Honoraria; TG Therapeutics: Honoraria; Medison Ventures: Honoraria; Merck: Honoraria; Gilead: Honoraria; BMS: Honoraria; Roche: Honoraria, Research Funding; Seattle Genetics: Honoraria. Crump: Roche: Research Funding; Epizyme: Research Funding; Kyte/Gilead: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees. Prica: Kite Gilead: Honoraria; Astra-Zeneca: Honoraria. Chen: Beigene: Membership on an entity's Board of Directors or advisory committees; Astrazeneca: Membership on an entity's Board of Directors or advisory committees; BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Gilead: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy. Kridel: Gilead Sciences: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal